Life Sciences Translation Company
Sesen helps life sciences teams manage clinical, regulatory, labeling, and AI-enabled translation workflows across 150+ languages.
Services
Life Sciences Language Services
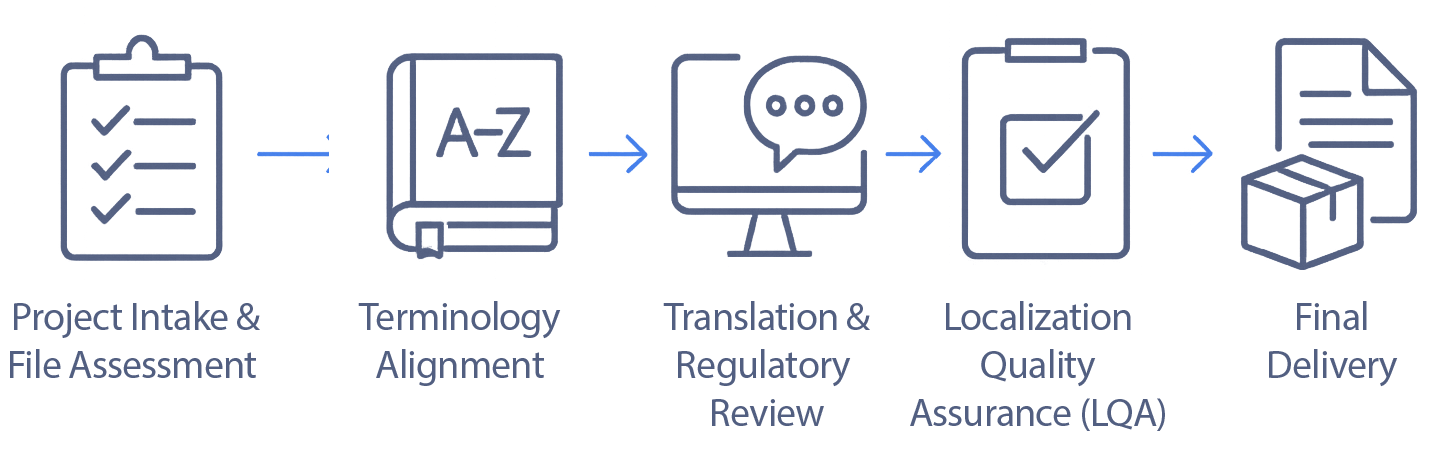
Specialized translation and localization for regulated life sciences content.
Explore Sesen services for clinical, regulatory, labeling, medical device, software, multimedia, and global commercial content.
Featured Services
Most Requested
Clinical Trial Translation
ICFs, protocols, site documents, and patient-facing materials.
Regulatory Translation & Compliance
Submissions, SOPs, audits, and regulated documentation.
Drug Labeling & Packaging Translation
Labeling, packaging, and product content for global markets.
IFU Translation Services
Instructions for use and medical device documentation.
More Services
Linguistic Validation
COA, PRO, questionnaire, and instrument workflows.
Medical & Scientific Translation
Specialized translation for medical and scientific content.
Clinical & Medical Software Localization
Software, portals, apps, and connected health interfaces.
Multimedia Localization
Video, voiceover, subtitles, and training content localization.
Interpreting Services
Remote and onsite interpreting for life sciences settings.
Marketing & Commercial Translation
Commercial, launch, and market access content for global audiences.
Need Help Choosing?
Talk with Team Sesen
Discuss clinical, regulatory, labeling, or AI-enabled translation needs with a life sciences specialist.
Life Sciences
Life Sciences
Specialized multilingual support across the life sciences ecosystem.
Explore Sesen support for pharmaceutical, biotechnology, medical device, CRO, healthcare, public health, and regulated content teams.
Core Life Sciences Sectors
Industry Focus
Healthcare & Public Sector
Specialized Domain Expertise
Language support aligned with how life sciences teams manage global content.
✓Clinical Development
Study startup, patient materials, clinical documentation, and global trials.
Regulatory Affairs
Submissions, labeling updates, safety documents, and compliance workflows.
Medical Affairs
Scientific exchange, publications, training, and medical communications.
Product Lifecycle & Market Access
Labeling, packaging, launch materials, updates, and commercialization.
Trust & Compliance
Built for regulated life sciences content.
- ✓ISO 17100, ISO 9001, and ISO 13485 certified
- ✓Professional native medical linguists
- ✓AI-enabled workflows with expert human review
- ✓AWS-hosted infrastructure
- ✓Support across 150+ languages
Plan a Global Program
Discuss Your Life Sciences Translation Needs
Connect with Team Sesen to review clinical, regulatory, labeling, or AI-enabled multilingual workflow needs.
Enterprise solutions for regulated multilingual content.
Explore structured multilingual workflows for clinical, regulatory, labeling, commercial, training, and patient engagement programs.
Structured Workflows
Regulatory Translation & Compliance
Global submissions, labeling audits, SOPs, and regulated documentation.
Clinical Trial Enablement
ICFs, protocols, site documents, patient materials, and recruitment content.
Labeling Workspace
NEWStructured workflows for labeling translation, validation, and approval.
Label Review & In-Context QA
In-layout linguistic QA for packaging, IFUs, labeling, and compliance.
Pharmacovigilance Translation
AE and SAE reports, DSURs, safety updates, and surveillance content.
Modular Submission & Content Reuse
Component-based translation for IDMP, SPL, and XML content workflows.
Global Engagement
Global Product Launch & Market Entry
Support for launching products across international markets.
Patient Engagement & Recruitment
Culturally adapted outreach and multilingual trial communications.
Terminology Management & Harmonization
Consistent scientific and brand language across global markets.
eLearning & Training Localization
Localized training for internal teams, HCPs, and external stakeholders.
Website & Digital Experience Localization
Websites, portals, mobile health apps, and digital patient experiences.
AI-enabled workflows for multilingual regulated content.
Sesen combines expert linguists, validation-driven review, and AI-assisted technologies to support global life sciences operations.
EXPLORE SESENGPTBuild a Regulated Multilingual Workflow With Sesen
Connect with Team Sesen to discuss clinical, regulatory, labeling, commercial, or AI-enabled multilingual operations.
AI & Innovation
AI
Powered by SesenGPT workflowsAI infrastructure for multilingual life sciences content.
Explore Sesen AI workflows for translation, terminology, validation, labeling, clinical trials, regulatory content, safety, and digital health localization.
Core AI Platform
Platform Capabilities
SesenGPT
COREAI-assisted translation workflows built for regulated life sciences content.
Hybrid Translation Workflows
Human-reviewed AI translation for quality, consistency, and scalability.
AI Validation & QA
Automated checks for terminology, numbers, formatting, and compliance.
Terminology Intelligence
AI-assisted term extraction, harmonization, and reuse across global content.
Labeling Workspace
NEWStructured workflows for multilingual labeling translation, review, and validation.
Operational AI Solutions
Workflow Applications
Clinical Trial AI Workflows
AI-assisted multilingual support for ICFs, protocols, and patient materials.
Regulatory Content Automation
Structured AI workflows for submissions, labeling, and compliance content.
Pharmacovigilance AI Support
AI-enabled workflows for AE reports, DSURs, safety updates, and drug surveillance.
Digital Health AI Localization
AI-assisted localization for portals, apps, eCOA, and digital health platforms.
AI Workflow Orchestration
Coordinated multilingual workflows with AI routing and human review.
Controlled AI Translation Program
Build a controlled AI translation program.
Start with a pilot that combines SesenGPT, professional native medical linguists, terminology control, and validation-driven QA.
EXPLORE SESENGPTDiscuss AI Workflows
Plan AI-Enabled Multilingual Operations With Sesen
Connect with Team Sesen to discuss AI-assisted translation, terminology governance, multilingual QA, and regulated life sciences workflows.
Resources
Resources
Explore all resourcesInsights and knowledge resources for life sciences teams.
Access expert perspectives, proof points, learning resources, and practical tools for multilingual life sciences content programs.
Industry Insights
Expert Perspectives
Blog & Insights
Trends, analysis, and expert perspectives on multilingual life sciences communication.
AI & Regulatory Insights
AIGuidance on AI-enabled workflows, validation, terminology, and compliance.
Clinical & Regulatory Knowledge
Best practices for clinical trials, submissions, labeling, and global content operations.
Customer & Operational Resources
Proof Points & Tools
Case Studies & Success Stories
Examples of multilingual workflow support across life sciences organizations.
Webinars & Events
On-demand sessions and expert discussions on regulated multilingual operations.
Glossaries & Style Guides
Terminology resources for consistency across multilingual content programs.
Strategic & Technical Resources
Guidance Library
AI Translation Guidance
Explore guidance for regulated AI translation workflows.
Review insights on validation, terminology, structured content, compliance, and multilingual operations for life sciences organizations.
Explore AI InsightsResource Center
Find the Right Resource for Your Team
Explore Sesen resources for clinical, regulatory, AI-enabled, quality, and multilingual content operations.
People, values, and global presence behind regulated multilingual content.
Learn about Sesen's mission, leadership, values, global footprint, partners, and commitment to life sciences localization excellence.
Company Foundation
Mission & Leadership
Global Presence
Locations & Updates
Why Teams Work With Sesen
Built around regulated content.
Sesen combines life sciences specialization, multilingual operations, and AI-enabled workflows for organizations that need quality, consistency, and scale.
Talk With Team Sesen
Discuss Enterprise Translation and Localization Needs
Connect with Team Sesen to discuss enterprise translation, localization, or AI-enabled workflow needs.
Contact
Contact
Talk with our teamConnect with Sesen's life sciences translation team.
Choose the right path for a new translation, localization, AI-enabled workflow, partnership, or existing project support need.
Start a Project
New Translation Needs
Request a Translation Quote
Get pricing and turnaround estimates for regulated multilingual content.
Schedule a Consultation
Speak with our team about clinical, regulatory, labeling, or AI-enabled workflows.
Start an AI Pilot Program
AIEvaluate SesenGPT workflows for multilingual regulated content.
Enterprise Engagement
Sales, Partnership & Support
Why Organizations Contact Sesen
Trusted for clinical, regulatory, and product content.
- ✓ Clinical trial translation
- ✓ Regulatory submissions
- ✓ Labeling workflows
- ✓ Pharmacovigilance support
- ✓ AI-enabled multilingual operations
General Contact Information
Need Help Finding the Right Contact Path?
For general inquiries, support, and company information, connect with Team Sesen and we will route your request to the right team.